Organic Chemistry Laboratory II
Comparison of Substitution and Elimination Reactions
Experimental Procedure
|
|
In this two-week
experiment, students will work in pairs to react an
alkyl bromide (substrate) with and alkoxide
(nucleophile or base) to determine the ratio of
substitution products (ether) relative to elimination
products (alkene(s)). Each bench in the lab will
be assigned specific reactions (see table below) and
students at each bench will work in pairs to complete
the two assigned reactions for that bench. The
product(s) from each reaction will be analyzed by TLC,
chemical tests, and gas chromatography-mass
spectrometry (GC-MS).
Students will preform TLC analysis
and chemical tests. The GC-MS data will be provided. The effect
of alkyl bromide substitution and structure of the
alkoxide will be determined by compiling results from
all of the groups.
|
|
| Bench |
Group |
Reaction |
|
Bench |
Group |
Reaction |
| A |
1 |
1-bromopentane
+ sodium methoxide |
D |
1 |
2-bromo-2-methylbutane
+ sodium methoxide |
|
| 2 |
2-bromopentane
+ sodium methoxide |
2 |
2-bromo-2-methylbutane + sodium ethoxide | |||
| B |
1 |
1-bromopentane
+ sodium ethoxide |
E |
1 |
2-bromo-2-methylbutane
+ potassium tert-butoxide |
|
| 2 |
2-bromopentane
+ sodium ethoxide |
2 |
1-bromopentane +
potassium tert-butoxide |
|||
| C |
1 |
1-bromopentane
+ potassium tert-butoxide |
F |
1 |
2-bromopentane + sodium methoxide | |
| 2 |
2-bromopentane
+ potassium tert-butoxide |
2 |
1-bromopentane + sodium ethoxide | |||
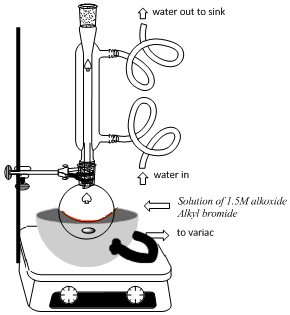
|
Reaction
Clamp a round-bottomed (rb) reaction flask (25ml) to the vertical bar in the hood. Place heating mantle (50ml) on a stir plate and position the heating mantle around the bottom of the rb flask. Add a stir bar to the flask. Measure out 1.2ml of the alkoxide (1.5M, NaOMe or NaOEt or KOt-Bu) using a 10ml graduated cylinder and add the alkoxide to the flask using a powder funnel, being careful not to drip the solution on the flask joint. Add the alkyl bromide (10 drops, ~0.5ml, ~ 4.0 mmol) to the reaction flask. Attach a reflux condenser to the rb flask and insert a drying tube at the top of the reflux condenser. Connect the water to the condenser tube, water in from the bottom, out from the top. Connect the heating mantle to a Variac, turn on the water, start stirrer, and begin heating the reaction mixture (See figure at left). Continue heating at reflux for one hour. After 1 hour, cool the reaction mixture to room temperature and transfer the reaction mixture to a 125ml separatory funnel, positioned in a ring, attached to a ring stand or the bars in the hood (See figure at right). Add MTBE (10ml) to the reaction flask, swirl to wash remaining reaction product and add the MTBE solution to the separatory funnel using a powder funnel. Add saturated aqueous ammonium chloride (10ml) to the separatory funnel, stopper the separatory funnel, shake and drain the aqueous layer into the 50ml beaker labeled "aqueous layer". (Be sure to remove the stopper to allow the liquid layers to drain from the separatory funnel.) Add an additional 10mL of saturated aqueous ammonium chloride to the separatory funnel, shake drain the aqueous layers into the same beakes. Repeat this step one last time using water (10mL) to wash the organic layer. Drain the water layer into the same aqueous layer beaker and the final organic layer into the beaker labeled "organic layer". Dry the organic layer with anhydrous magnesium sulfate and decant the dried organic layer into a clean, dry Erlenmeyer flask or beaker (25 or 50ml). |
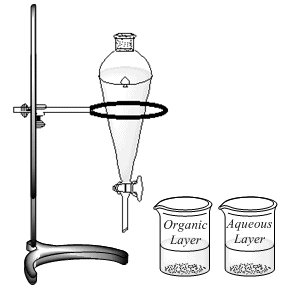 |
| Chemical Tests Run all four of the chemical tests listed in the table below on your starting material, alkene standard(s)(from the table below) and reaction mixture. Click on the link for the procedure for each test. |
|
 |
| Chemical
Test |
Description |
| Sodium
Iodide in Acetone |
This is a chemical test for primary and secondary alkyl bromides. Tertiary alkyl bromides and sometimes secondary alkyl bromide, react slowly or not at all. Ethers and alkenes do not react. |
| Silver
Nitrate in Ethanol |
This is a chemical test, primarily for tertiary alkyl bromides. Tertiary alkyl bromides react relatively quickly compared to secondary and primary alkyl bromides. Ethers and alkenes do not react. Occasionally, secondary and primary alkyl bromides give false positive results. |
| KMnO4
Test |
This is a chemical test
for alkenes. Ethers and alkyl bromides do not
react |
| Bromination
Test |
This is a test for alkenes.
Ethers and alkyl bromides do not react. |
| Gas Chromatography-Mass
Spectrometry Gas chromatograms for the reaction mixture for each reaction are provided below (Labeled as GC.A-1, GC.A-2 etc...). The mass spectrum for each peak in each reaction chromatogram are also given in the corresponding column in the table below. Use this data to determine the reaction products that were generated in your reaction and if the starting material (or most of the starting material) was converted to one or more reaction products, and the major product(s) of the reaction. Each of the gas chromatograms from the reactions that are done have two or more major peaks. Each peak in the chromatogram corresponds to the starting material (the alkyl bromide) and/or one or more reaction products. The reaction product(s) may be generated through an SN2 mechanism to form an ether (alkoxide acts as a nucleophile) and/or one or more alkene may form via an E2 mechanism, where the alkoxide acts as a base. The alkene product(s) that form(s) will be dependent on the structure of the starting alkyl bromide. The area of each peak in the chromatogram correlates to the amount of that compound in the reaction mixture and the peak area ratios are provided in the table below. The larger the peak area, the more of that compound is present. These peak areas can be used to determine the major product(s) of the reaction as well as the ratio of SN2 versus E2 products. Mass spectra are provided for each of the peaks that appear in the corresponding gas chromatograms. The mass spectra are used to identify the compounds in the reaction mixture. The alkyl bromide starting material has a mass spectrum (and molecular weight) that is distinctly different from the SN2 and E2 product(s). Likewise, the SN2 product has a mass spectrum that is uniquely different from the E2 product. Using the mass spectrum for each peak in the chromatogram, you can determine the molecular weight of each compound in the reaction mixture, and thus the identity of each compound. When combined with the peak areas, the major reaction product(s) can be determined as well as a ratio of SN2 versus E2 products. Before you leave the lab, provide the lab instructor with the name(s) of the major product(s) of your reaction. |
| Bench |
Group |
Gas Chromatogram |
Mass Spectra/GC
Peak Ratio |
|
Bench |
Group |
Gas Chromatogram |
Mass Spectra/GC
Peak Ratio |
| A |
1 |
GC.A-1 |
Peak 1
85% Peak 2 15% |
D |
1 |
GC.D-1 |
Peak 1
16% Peak 2 35% Peak 3 52% |
|
| 2 |
GC.A-2 |
Peak 1
11% Peak 2 22% Peak 3 9% Peak 4 16% Peak 5 42% |
2 |
GC.D-2 |
Peak 1
10% Peak 2 40% Peak 3 20% Peak 4 30% |
|||
| B |
1 |
GC.B-1 |
Peak 1
20% Peak 2 80% |
E |
1 |
GC.E-1 |
Peak 1
55% Peak 2 33% Peak 3 12% |
|
| 2 |
GC.B-2 |
Peak 1
20% Peak 2 40% Peak 3 14% Peak 4 17% Peak 5 9% |
2 |
GC.E-2 |
Peak 1
20% Peak 2 27% Peak 3 53% |
|||
| C |
1 |
GC.C-1 |
Peak 1
20% Peak 2 27% Peak 3 53% |
F |
1 |
GC.F-1 |
Peak 1
11% Peak 2 22% Peak 3 9% Peak 4 16% Peak 5 42% |
|
| 2 |
GC.C-2 |
Peak 1
54% Peak 2 12% Peak 3 7% Peak 4 27% |
2 |
GC.F-2 |
Peak 1
20% Peak 2 80% |